Introduction
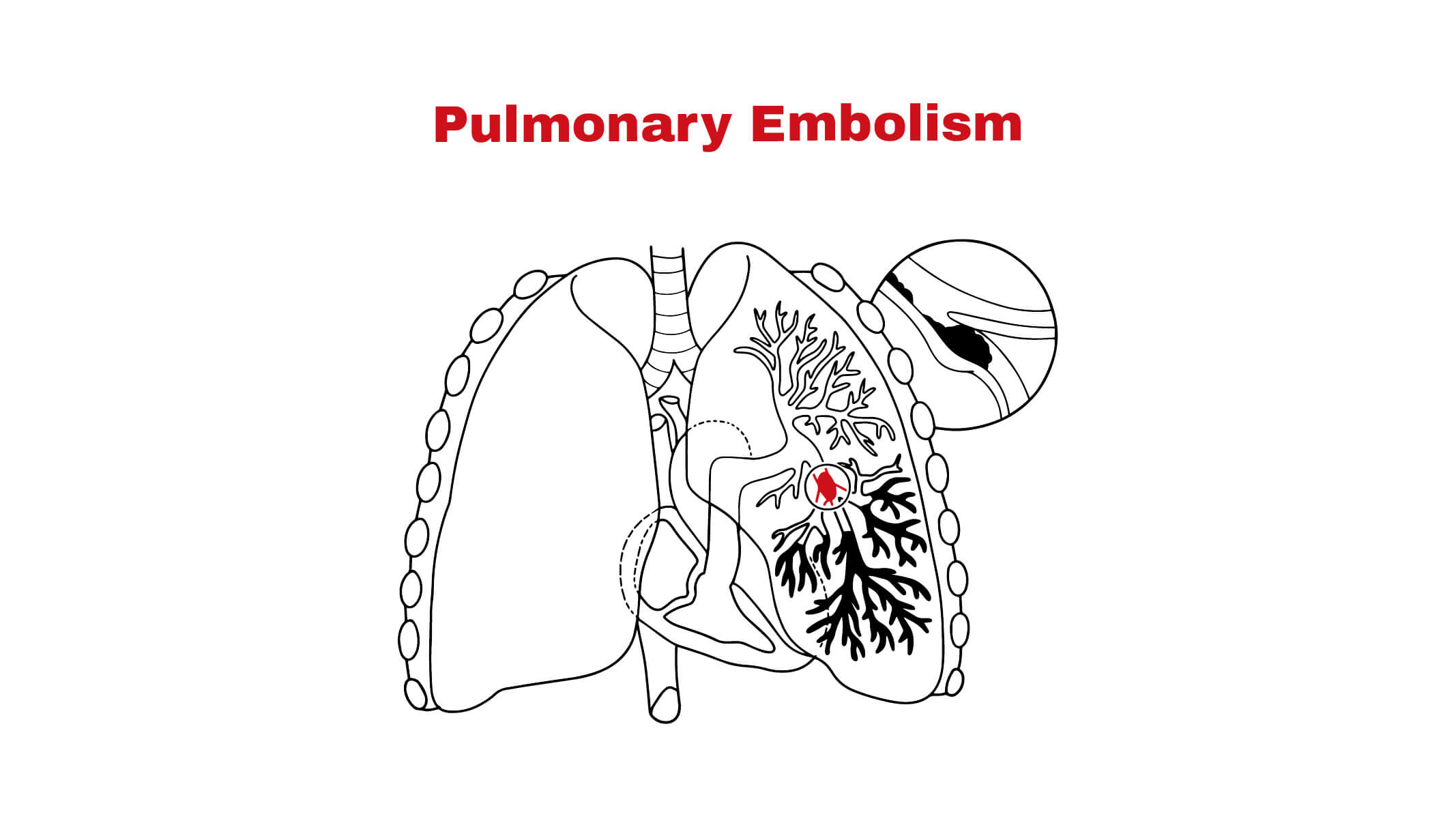
The recent decision by the NHS watchdog regarding the use of Tecartus, a CAR T-cell therapy for adult patients with relapsed or refractory B-cell malignancies, has sparked considerable discussion in the healthcare sector. The approval of this treatment is crucial as it offers a new avenue for patients who have limited options following conventional therapies. The significance of this decision lies in its implications for patient care, healthcare costs, and the future of cancer treatment in the UK.
Details of the Decision
The National Institute for Health and Care Excellence (NICE) made announcements in October 2023 regarding the evaluation of Tecartus as part of its ongoing assessment of cancer therapies. Tecartus, which is produced by Kite Pharma, has shown substantial efficacy in clinical trials, particularly in patients with mantle cell lymphoma. The watchdog’s decision comes after extensive data reviews, consultations with healthcare providers, and patient advocacy groups. This thorough evaluation aims to ensure that patients receive the best possible treatments while managing costs effectively for the NHS.
Implementation and Impact
If confirmed, the NHS’s approval will pave the way for Tecartus to be available for eligible patients across the country, marking a significant advance in targeted cancer therapies. The implications of this decision extend beyond just the patient spectrum; they influence research funding, healthcare policy, and resource allocation across the NHS. As the healthcare system grapples with increasing demands on its services, the inclusion of innovative treatments like Tecartus could lead to better patient outcomes and potentially lower long-term costs by reducing the need for hospital admissions and palliative care.
Conclusion
The Tecartus decision by the NHS watchdog is a pivotal moment for both patients with limited treatment options and the broader healthcare landscape in the UK. This approval, if implemented, could enhance the quality of life for many undergoing treatment for B-cell malignancies. As the case of Tecartus unfolds, it serves as a marker of the balance between innovation in healthcare and the pragmatic financial considerations that guide treatment approvals. Stakeholders are keenly observing how this decision shapes the future of oncology treatment in the NHS and expectations from upcoming therapy evaluations.